

ISO 13485 is designed to be used by organizations involved in the design, production, installation and servicing of medical devices and related services. It can also be used by internal and external parties, such as certification bodies, to help them with their auditing processes.


ISO 13485 is the internally recognized gold standard for quality in the medical device industry. Certification to this standard shows clients and customers that your company takes quality very seriously, and that you have a system in place to ensure it. Your QMS can be a powerful marketing tool, and it has become a requirement in many countries for suppliers to show compliance. This translates to more opportunity.
The ISO 13485 standard is built on a set of quality management principles, one of which is ensuring customer satisfaction. This can be achieved by assessing customer needs and expectations, and striving to meet them. Customers know what they want, and what they need, and many of them will not even entertain a supplier that isn’t certified. Beyond that, ensuring the satisfaction of your existing customers keeps them coming back, and helps you sell your services to new customers. This translates directly to increased revenue.
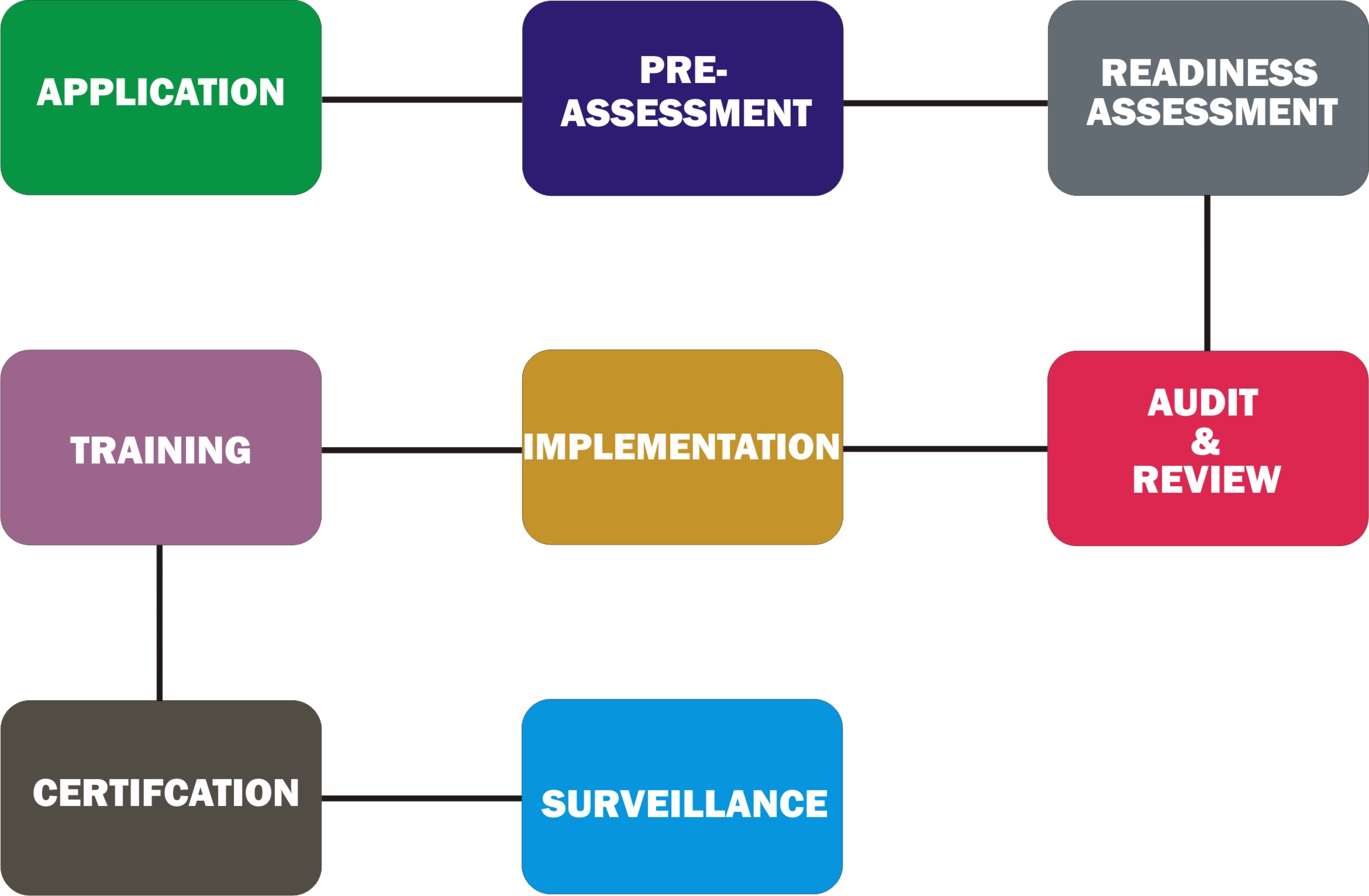
These are some of the items that may be required for the ISO certification process, assessors may require more but your company need to have this basic information ready
Note* - that in case you do now have any of the listed information, our assesors and consultants will work with you to put them in place. You may still go ahead with your application.
